You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Tissue Culture - Umami Style
- Thread starter Umamiseedco
- Start date
Rico D
Active member
it is very challenging and takes a lot of practice and patience. I got to try this hands on and the learning curve is steep. Even the transition from working with your eyes directly on an object vs working on a screen was a challenge. But like anything else practice makes perfect.
It does seem like most advertise/show off shoot tc vs meristematic. Never got that.
lastly, why hasnt anyone brought sour d back to life yet!?!?
It does seem like most advertise/show off shoot tc vs meristematic. Never got that.
lastly, why hasnt anyone brought sour d back to life yet!?!?
Umamiseedco
Well-known member
Sour is very much alive and well in my greenhouse. I just dusted her with zeclair and jelly zonuts!it is very challenging and takes a lot of practice and patience. I got to try this hands on and the learning curve is steep. Even the transition from working with your eyes directly on an object vs working on a screen was a challenge. But like anything else practice makes perfect.
It does seem like most advertise/show off shoot tc vs meristematic. Never got that.
lastly, why hasnt anyone brought sour d back to life yet!?!?
Rico D
Active member
Sour is very much alive and well in my greenhouse. I just dusted her with zeclair and jelly zonuts!
Jive Genetics
New member
Really good info in this thread, much appreciated. Looking forward to popping your beans and hopefully finding some stock to breed with in the future! TC is quite a ways down the line for me but I think it will come in handy when I have the bandwidth to do so. Any suggested reading material for 101 level TC knowledge? I got the book Plants In Test Tubes and my dog ate it up before I could comb it over 
Anthem27.5
Member
This is great information thank you for sharing!In this example, you can actually see the virus as it moves up the stem. The virus grows just as fast as the plant cells do, so you are trying to "chase out" the infected cells.
View attachment 357025
Umamiseedco
Well-known member
read this: https://summit.sfu.ca/_flysystem/fedora/2022-11/etd21287.pdfReally good info in this thread, much appreciated. Looking forward to popping your beans and hopefully finding some stock to breed with in the future! TC is quite a ways down the line for me but I think it will come in handy when I have the bandwidth to do so. Any suggested reading material for 101 level TC knowledge? I got the book Plants In Test Tubes and my dog ate it up before I could comb it over
and watch a bunch of youtube videos.
Umamiseedco
Well-known member
Umamiseedco
Well-known member
Rumpelstiltskin
Walking on Water
Its great to see threads of this nature...finally. What experience do you have using turmeric and benzoin(sambrani) resin-based fumigation methods for microbial contamination surface sterilization of explants as opposed to using chemicals that become toxic to explants and surfaces as doses increase?
Umamiseedco
Well-known member
Honestly, none. I have had great success with iodine + soap and then varying levels of chlorine for keeping explants sterile. The biggest issue that I have faced is endophyte contamination. I seem to have alleviated that for the most part by running a sterile media and fertilizer. Also, I don't recommend using PPM (plant preservation mixture) for general use, but it does work pretty well for endophyte issues.Its great to see threads of this nature...finally. What experience do you have using turmeric and benzoin(sambrani) resin-based fumigation methods for microbial contamination surface sterilization of explants as opposed to using chemicals that become toxic to explants and surfaces as doses increase?
Umamiseedco
Well-known member
Umamiseedco
Well-known member
Umamiseedco
Well-known member
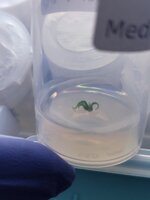
So lets check out what we got after another week of lab work. Zeclair is looking great for one weeks worth of growth!

Payzos is also looking nice and healthy in stage 1.


This far, my contamination rate is less than 1%. Hopefully that will continue but endophytes have a habit of popping up when you least expect them... Sometimes as far as a few months into the process. Meristem cultures (as shown here) are usually immune, but It does happen resulting in a loss of individual explants.
When the contamination does occur, I remove the other healthy explants and transplant them into fresh media and hope for the best.


Payzos is also looking nice and healthy in stage 1.


This far, my contamination rate is less than 1%. Hopefully that will continue but endophytes have a habit of popping up when you least expect them... Sometimes as far as a few months into the process. Meristem cultures (as shown here) are usually immune, but It does happen resulting in a loss of individual explants.
When the contamination does occur, I remove the other healthy explants and transplant them into fresh media and hope for the best.

Umamiseedco
Well-known member
with these deli lids yes. They seal like crazy well and are super hard to remove without pinching the container. The pop tops on the other hand can get a little wonky and don't always stay shut.have you found taping the lids unnecessary with your containers?
kushyman
Well-known member
for sterilization how about HOCL?Honestly, none. I have had great success with iodine + soap and then varying levels of chlorine for keeping explants sterile. The biggest issue that I have faced is endophyte contamination. I seem to have alleviated that for the most part by running a sterile media and fertilizer. Also, I don't recommend using PPM (plant preservation mixture) for general use, but it does work pretty well for endophyte issues.
my lettuce raft has never looked better......
Umamiseedco
Well-known member
Any free available chlorine (FAC) will work just fine. I make mine from calcium hypochlorite at 5000ppm chlorine, and the active chemical from that mix is HOCL. You can use bleach as well, hypochlorite (OCl-). Both are effective at disinfection, but chemically speaking – hypochlorous acid is most effective for this purpose.
kushyman
Well-known member
we use the electric kettle like machine ..distilled water and salt..hypochlorous acid is most effective for this purpose.
kushyman
Well-known member
5000..<--interesting.Any free available chlorine (FAC) will work just fine. I make mine from calcium hypochlorite at 5000ppm chlorine, and the active chemical from that mix is HOCL. You can use bleach as well, hypochlorite (OCl-). Both are effective at disinfection, but chemically speaking – hypochlorous acid is most effective for this purpose.
the hocl plant Info of I have read says they start at under 50 ppms
and that 5 ppms is good enough.[.yeh right..maybe for a reservoir].[.jk]
..of course we like to push things as far as we can....[for science]